|
Now, let us determine the moles of each (I'll skip typing the calcs):ġ) Determine mass of all four compounds in the 40.10 g sample: In which case, the percentages above become grams. Oxygen: we leave this to later (see below for an interesting solution path that involves determining the mass of oxygen.)Ĭhlorine: problem gives 22.06 g in 75.00 g sampleĢ) Let us determine the percent composition: Another sample of the compound with a mass of 75.00 g is found to contain 22.06 g of Cl. Problem #4: The combustion of 40.10 g of a compound which contains only C, H, Cl and O yields 58.57 g of CO 2 and 14.98 g of H 2O. What is menthol's empirical formula? (Yes, the answer will be the same as #12.)   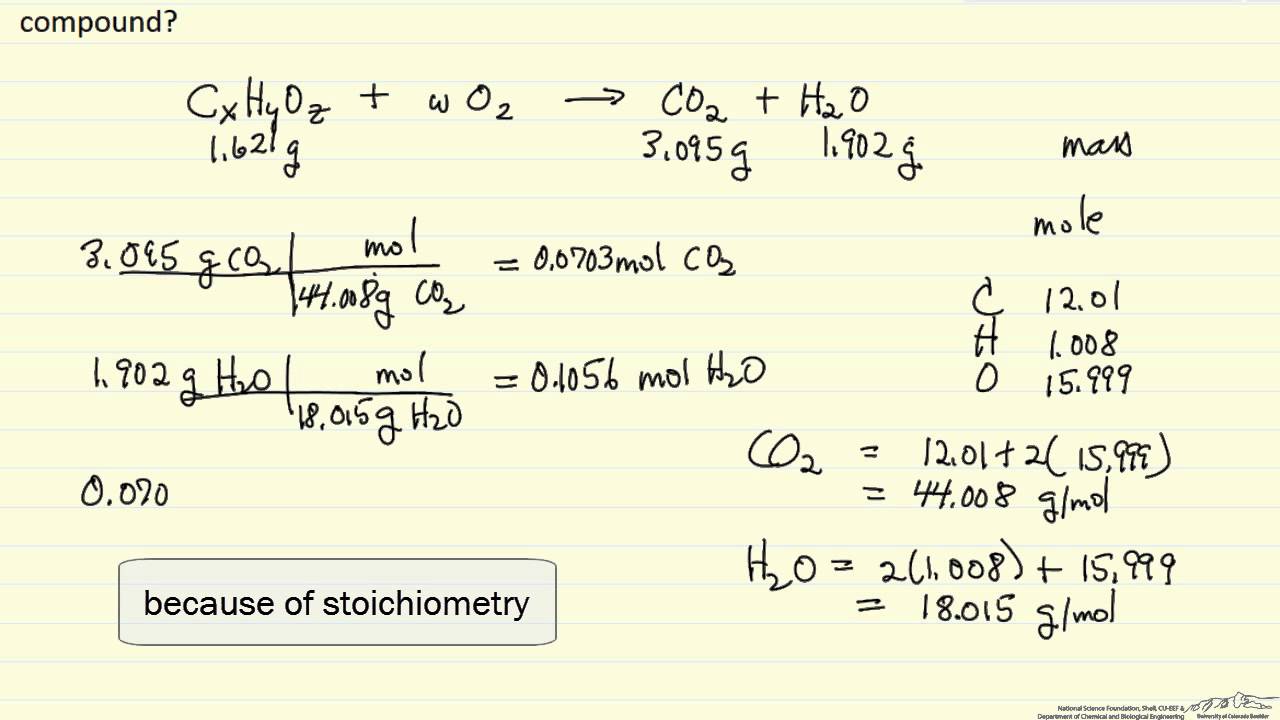
Problem #3: 0.1005 g of menthol is combusted, producing 0.2829 g of CO 2 and 0.1159 g of H 2O. Problem #2: 95.6 mg of menthol (molar mass = 156 g/mol) are burned in oxygen gas to give 269 mg CO 2 and 110 mg H 2O. Determine the empirical and molecular formulas. Problem #1: 0.487 grams of quinine (molar mass = 324 g/mol) is combusted and found to produce 1.321 g CO 2, 0.325 g H 2O and 0.0421 g nitrogen. Go to a discussion of empirical and molecular formulas. ChemTeam: Combustion Analysis: Problems 1 - 10 Combustion Analysis: Problems 1 - 10
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
